Federal Judge Denies Block on Mail-Order Abortion Pill but Orders FDA Review Completion
A U.S. federal judge in Louisiana ruled on Tuesday that the Food and Drug Administration must complete its review of mifepristone mailing restrictions, denying a preliminary block for now. The decision maintains national access to the abortion pill via mail and telehealth temporarily. The ruling stems from a lawsuit challenging FDA policies on the drug used in medication abortions.
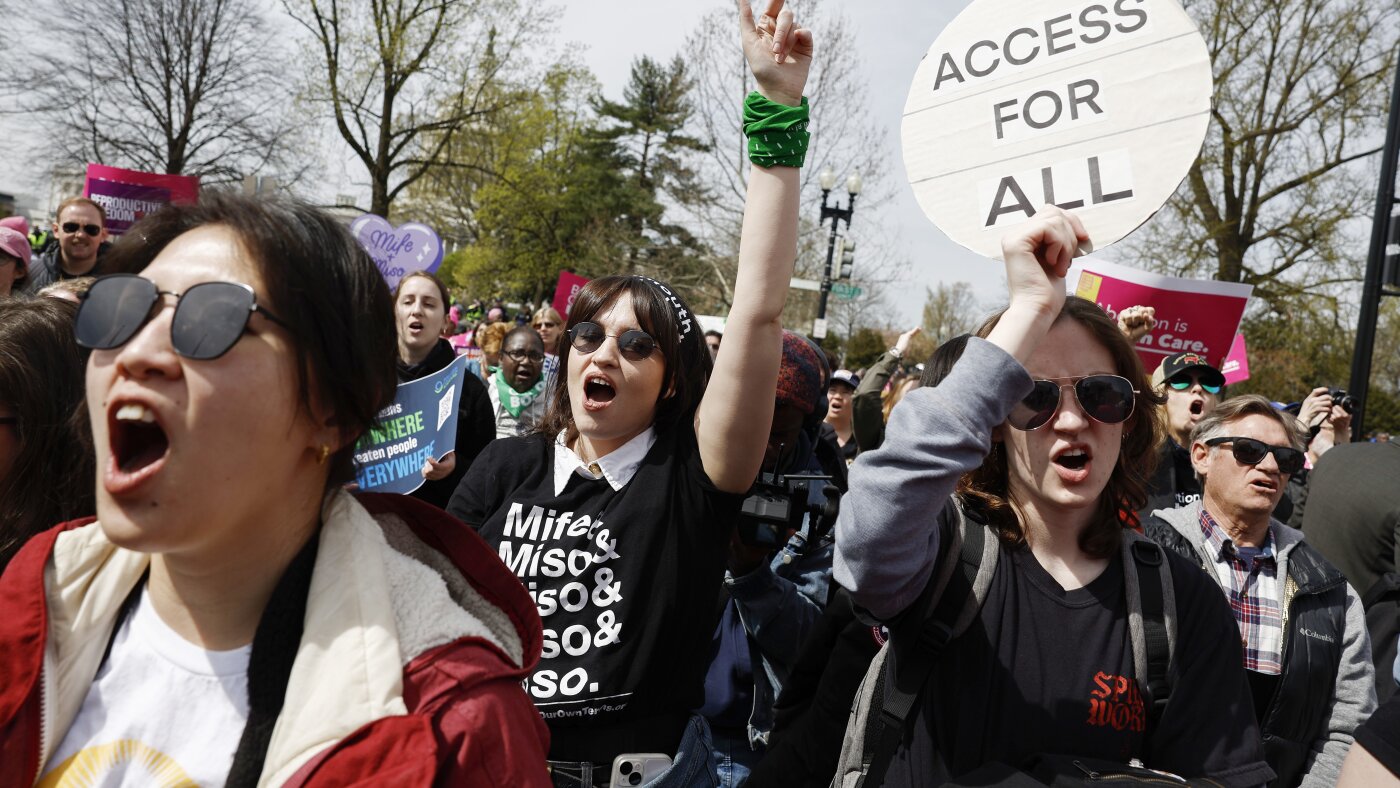 NPR
NPRA federal judge in the Western District of Louisiana ruled on Tuesday that access to mifepristone, a drug used in medication abortions, remains unchanged nationally for the time being. The decision denies a request to immediately block the FDA's policy allowing the pill to be sent by mail and obtained through telehealth. Judge David C.
Joseph issued a 37-page opinion emphasizing that the FDA must finish its ongoing review of the policy. The lawsuit was filed by plaintiffs seeking to restrict mifepristone distribution, arguing that mailing the drug poses safety risks. Judge Joseph found insufficient evidence at this stage to halt the current access methods.
The ruling applies nationwide, preserving telehealth abortion options amid ongoing legal challenges.
his opinion, Judge Joseph stated that the FDA's review process, initiated in response to prior litigation, requires completion before any changes can be imposed.
The judge rejected the plaintiffs' motion for a preliminary injunction, noting that the agency's evaluation addresses the core concerns raised. This decision follows a series of court battles over mifepristone since its approval decades ago. The FDA approved mifepristone in 2000 for use in combination with misoprostol to terminate pregnancies up to 10 weeks gestation.
Recent lawsuits have targeted expansions in access, including mail delivery approved in 2021 during the COVID-19 pandemic. Judge Joseph's order mandates the FDA to finalize its review within specified timelines, though exact dates were not detailed in available reports.
abortions have increased significantly since 2020, with mifepristone accounting for over half of U.S. abortions. The ruling comes after the Supreme Court's 2024 decision upholding FDA authority on the drug's approval, but it leaves room for further restrictions pending the review.
Advocacy groups on both sides monitor the case closely, as it could affect access in states with varying abortion laws.
NPR reported that the judge's opinion is far from an endorsement of telehealth abortion, highlighting reservations about the method's expansion. The case is part of wider litigation, including challenges in Texas and other districts, where similar issues are under consideration. No immediate changes to prescription or distribution practices were ordered.
Key Facts
Story Timeline
4 events- Tuesday, October 8, 2024
Judge David C. Joseph issues 37-page ruling denying preliminary injunction on mifepristone mail access.
2 sourcesNPR · STAT News - 2021
FDA approves mail delivery of mifepristone in response to COVID-19 pandemic needs.
2 sourcesNPR · AP - 2000
FDA initially approves mifepristone for use in medication abortions up to 10 weeks.
2 sourcesNPR · AP - June 2024
Supreme Court upholds FDA's approval of mifepristone in related case.
1 sourceNPR
Potential Impact
- 01
Telehealth abortion services continue operating without immediate restrictions nationwide.
- 02
Access to medication abortion expands in states without total bans.
- 03
Plaintiffs pursue appeals to overturn the denial of injunction.
- 04
FDA completes review of mifepristone mailing policy by mandated deadline.
Transparency Panel
Related Stories
 deccanchronicle.com
deccanchronicle.comHantavirus Cases Reported on MV Hondius Cruise Ship, Three Fatalities Amid Low Transmission Risk
A hantavirus outbreak on the MV Hondius cruise ship has killed three passengers and sickened seven others, prompting an international response coordinated by the World Health Organization. The ship, carrying 147 people from 23 nationalities, is set to sail to Spain's Canary Islan…
 The U.S. Food and Drug Administration / Wikimedia (Public domain)
The U.S. Food and Drug Administration / Wikimedia (Public domain)FDA Commissioner Defends Drug Rejection Decisions
The FDA commissioner defended recent drug rejections in a CNBC interview, citing adherence to scientific reviews amid reports of agency turmoil. Criticism includes a high-profile denial of a melanoma treatment from Replimune and pressure from President Trump over vape approvals.…
 Prime Minister's Office / Wikimedia (GODL-India)
Prime Minister's Office / Wikimedia (GODL-India)Blackstone CEO Steve Schwarzman Hosts 10th Reunion for Schwarzman Scholars Program in Beijing
Steve Schwarzman, CEO and Chairman of Blackstone, hosted 1,300 alumni at the 10th reunion of the Schwarzman Scholars program at Tsinghua University in late April. The event featured a keynote address and panel discussion amid reflections on U.S.-China relations. The program, insp…