Fifth Circuit Issues Temporary Nationwide Mifepristone Order
A U.S. appeals court issued an order on May 1, 2026, pausing FDA rules that allowed mifepristone to be obtained by mail or telemedicine. The decision, stemming from a Louisiana lawsuit, requires in-person dispensing nationwide. Stakeholders on both sides reacted, with the case likely headed to the Supreme Court.
 Npr
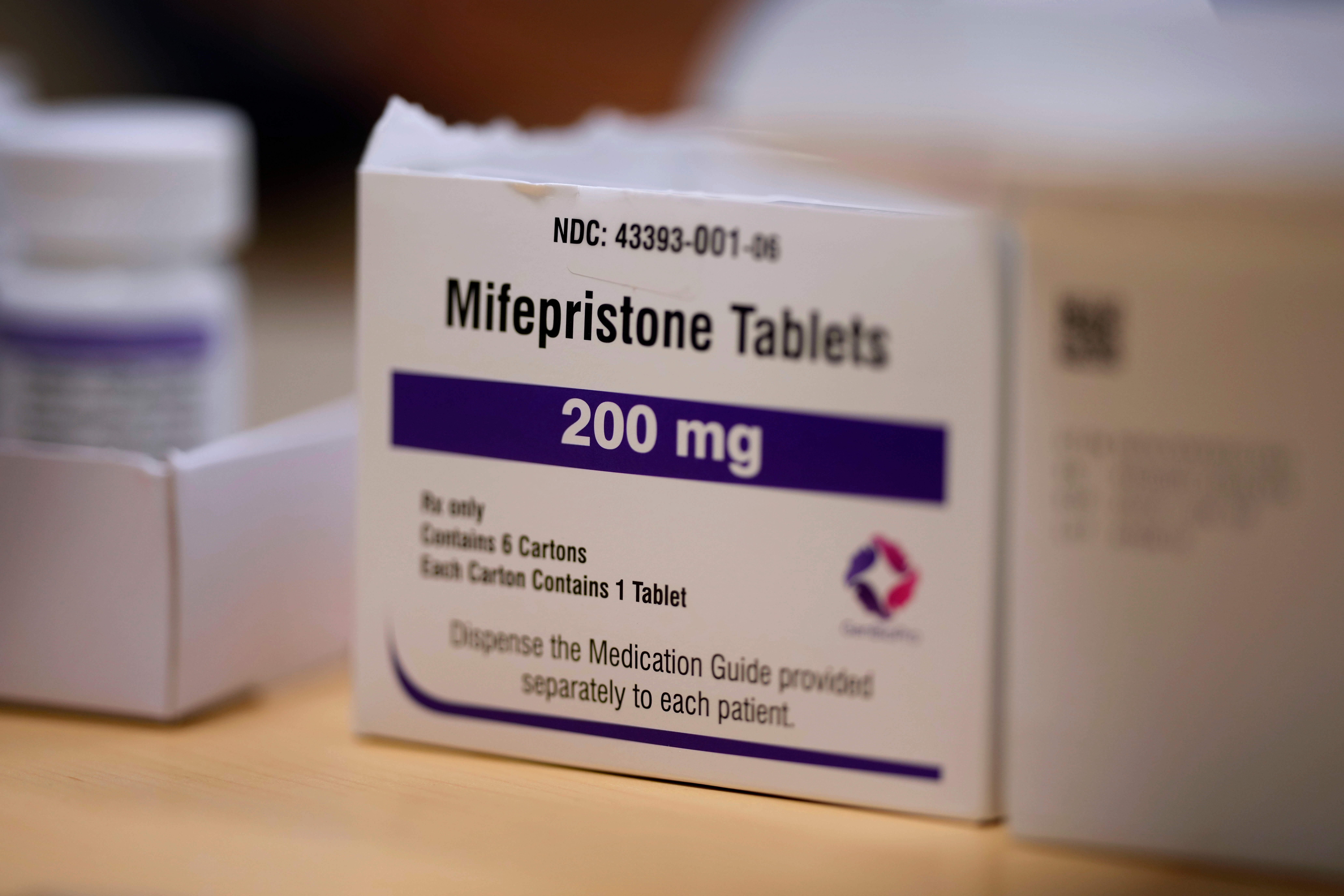
NprThe U.S. Court of Appeals for the Fifth Circuit issued an order on May 1, 2026, temporarily requiring that the abortion medication mifepristone be dispensed in person at clinics, hospitals, or medical offices, rather than through mail or telemedicine prescriptions.
Post by @ABC on X
The order, which has nationwide effect, stems from a lawsuit filed by the state of Louisiana challenging a 2023 Food and Drug Administration regulation that permitted mailing of the drug after virtual consultations.
" The court further noted that the ruling "protects fetal life consistent with state policy on personhood from conception," according to the filing. The decision overrides a lower court ruling from earlier in the month that had paused the case pending an FDA review directed by the Trump administration.
Mifepristone, approved by the FDA in September 2000 for terminating pregnancies up to seven weeks gestation (later extended to 10 weeks), blocks the hormone progesterone needed for pregnancy continuation. It is typically used in combination with misoprostol, with FDA data indicating the regimen is about 95% effective and requires follow-up care in less than 1% of cases.
The FDA temporarily lifted in-person dispensing requirements in April 2021 due to the COVID-19 pandemic and made the change permanent in 2023, citing over 20 years of monitoring and studies involving thousands of women showing safe use without direct supervision.
" Under the Trump administration, the agency announced it is conducting a new safety review of mifepristone at the president's direction, though it could not specify a completion date and noted it was still collecting data. The U.S. Supreme Court in 2024 unanimously rejected a separate challenge to mifepristone access, ruling that the anti-abortion doctors involved lacked legal standing to sue.
That decision followed the court's 2022 overturning of Roe v. Wade, which had established a nationwide right to abortion.
" Julia Kaye, an attorney with the American Civil Liberties Union, said the decision "defies clear science and settled law and advances an anti-abortion agenda that is deeply unpopular with the American people," adding that it would affect access to abortion and miscarriage care nationwide, particularly for rural communities, low-income individuals, people with disabilities, survivors of intimate partner violence, and communities of color.
" The American College of Obstetricians and Gynecologists has stated that mifepristone is one of the most common methods of abortion in the U.S. and has been used safely by millions over more than two decades. No public comments from the FDA on the specific order were available as of May 1, 2026, according to agency statements.
Key Facts
Story Timeline
7 events- May 2, 10:02 AM ET
3 new sources added: Abc News, Stat, The Independent
3 sourcesAbc News · Stat · The Independent - 2026-05-01
Fifth Circuit Court of Appeals issues order temporarily reinstating in-person requirement for mifepristone
2 sourcesThe BBC · NPR - 2024
US Supreme Court unanimously rejects effort to restrict access to mifepristone, ruling anti-abortion doctors lack standing
2 sourcesThe BBC · NPR - 2023
FDA permanently lifts in-person dispensing requirement for mifepristone, allowing mail delivery
2 sourcesThe BBC · NPR - 2022
US Supreme Court overturns Roe v. Wade, rescinding nationwide guarantee to abortion
2 sourcesThe BBC · NPR - 2021-04
FDA expands mifepristone availability by lifting in-person requirement during Covid-19 pandemic
1 sourceThe BBC - 2016
Approved use of mifepristone extended to 10 weeks of pregnancy
1 sourceThe BBC
Potential Impact
- 01
Continued legal abortion access in states like New York despite ruling
- 02
Reduced access to mifepristone in rural areas and for vulnerable groups via telemedicine
- 03
Potential appeal to Supreme Court, influencing nationwide abortion regulations
- 04
Effects on miscarriage care and broader reproductive health services
- 05
Ongoing FDA safety review under Trump administration may lead to further changes
Transparency Panel
Related Stories
 Trump White House Archived from Washington, DC / Wikimedia (Public domain)
Trump White House Archived from Washington, DC / Wikimedia (Public domain)Trump Pauses US Operation in Strait of Hormuz to Advance Iran Deal Talks
President Trump announced a temporary pause to Project Freedom, the U.S. effort to guide ships through the Strait of Hormuz, citing progress toward a settlement with Iran. The blockade on Iranian ports remains in effect, while Secretary of State Marco Rubio stated that the offens…
 foxnews.com
foxnews.comTom Homan Discusses Minneapolis Immigration Enforcement After Shooting
Tom Homan, White House Border Czar, stated that mass deportations can proceed in a smarter way. He acknowledged imperfections in the Minneapolis immigration crackdown following a deadly shooting. Homan highlighted efforts to address and fix issues after President Trump sent him t…
 Office of David Schweikert, US Congress / Wikimedia (Public domain)
Office of David Schweikert, US Congress / Wikimedia (Public domain)Canadian House Committee Hears Testimony on EV Import Deal with China
Industry experts and policy leaders testified before a Canadian parliamentary committee on the economic and security risks posed by a new trade agreement permitting low-tariff imports of Chinese electric vehicles. The deal, signed in January 2026, reduces tariffs and sets import…